Our work on mesoporous silicas and its applications has been accepted for publication in Microporous and Mesoporous Materials (impact factor 3.453) published by Elsevier. This journal is the official journal of International Zeolite Association. It covers novel and significant aspects of porous solids classified as either microporous (pore size up to 2 nm) or mesoporous (pore size 2 to 50 nm). The porosity should have a specific impact on the material properties or application. Typical examples are zeolites and zeolite-like materials, pillared materials, clathrasils and clathrates, carbon molecular sieves, ordered mesoporous materials, organic/inorganic porous hybrid materials, or porous metal oxides. Both natural and synthetic porous materials are within the scope of the journal.
Our accepted paper is:
PEG-templated mesoporous silicas using silicate precursor and their applications in desiccant dehumidification cooling systems
Heru Setyawan, Minta Yuwana, Ratna Balgis
HIGHLIGHTS
1. A cheap approach for producing mesoporous silica from silicate is proposed.
2. The mesoporous silicas have very high surface area which can reach > 600 m2/g.
3. The pore structure of mesoporous silicas can be tuned easily.
4. Impregnation of hygroscopic salt enhances significantly water adsorption.
5. The adsorbent composite is promising for desiccant dehumidifying cooling system.
Abstract
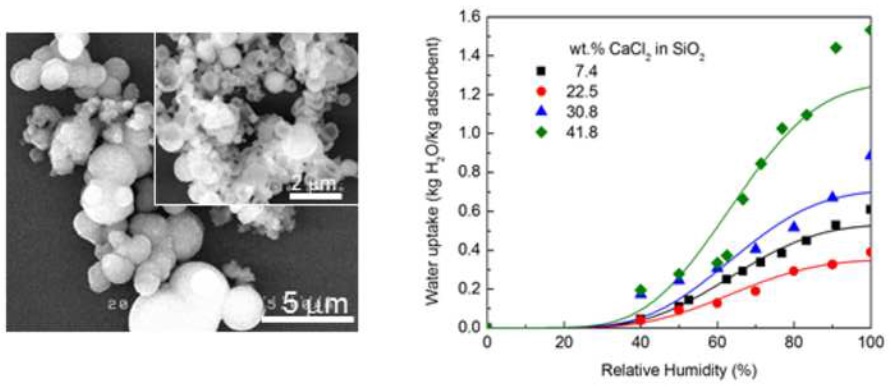
Polyethylene glycol (PEG) was employed as a templating agent for the preparation of mesoporous silica materials starting from sodium silicate solutions using a sol-gel method. We show that PEG can serve as a structure-directing agent resulting in silica structures with pores spanning a range of 3.3 to 3.9 nm depending on the PEG concentration and the PEG removal method. The use of a PEG template increased significantly the surface area from 252 m2 g-1 without PEG to >340 m2 g-1 and >634 m2 g-1 when the PEG was removed by, respectively, calcination and solvothermal extraction. It appears that calcination at high temperature caused a densification of the pore structure resulting in a smaller surface area and pore diameter. The impregnation of mesoporous silica with CaCl2 significantly increased the adsorption capacity of water. At high humidity, the composite adsorbents containing approximately 42 wt.% CaCl2 yield a maximum adsorption capacity of 1.6 times their own weight and 4.4 times that of bare silica. The adsorption is physical in nature as viewed from the adsorption energy calculated by the Dubinin-Radushkevich (D-R) equation.
Keywords: Mesoporous silica; Water Vapor; Sodium silicate; PEG-silica hybrid; Composite
adsorbent.


Leave a comment